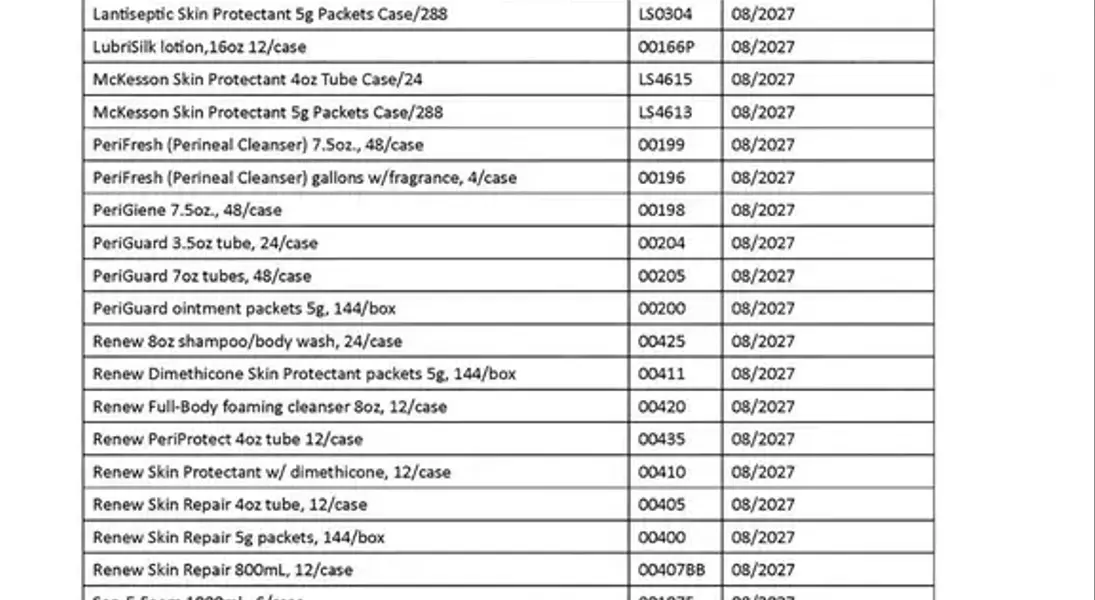

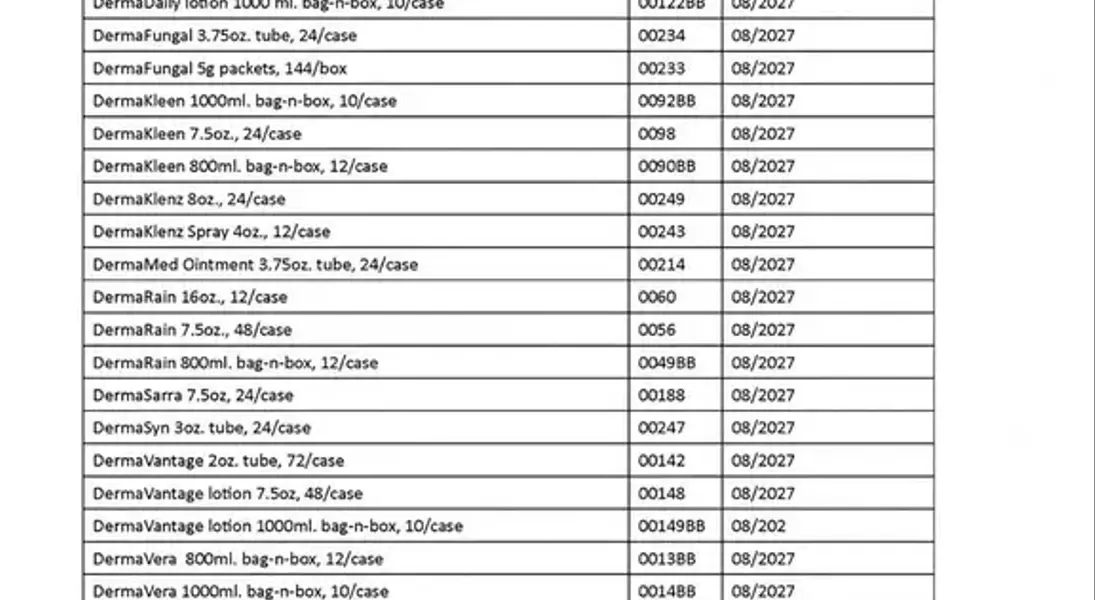


A critical public health announcement has been issued regarding a significant recall of numerous personal care and hygiene products, spanning across the entire nation. This widespread alert highlights the potential for microbial contamination, specifically involving the presence of Burkholderia cepacia bacteria, which poses a serious health risk to consumers. The affected products encompass a broad range of items, from hand sanitizers and cleansers to various ointments and lotions, all of which have been distributed and sold throughout the United States and Puerto Rico. This situation underscores the paramount importance of consumer vigilance in checking product labels and taking immediate action to safeguard personal health.
Nationwide Health Alert: Contaminated Topical Products Prompt Urgent Recall
In a crucial development for public safety, the U.S. Food and Drug Administration (FDA) has initiated a comprehensive nationwide recall affecting a diverse array of topical health and hygiene products. This includes numerous hand sanitizers, specialized cleansers, protective skin ointments, and deodorants. The genesis of this urgent action is the alarming discovery of potential contamination with Burkholderia cepacia, a bacterium known to cause significant health complications.
The recall extends to an extensive list of products, encompassing antifungal and moisturizing creams, distributed widely across the United States and Puerto Rico. Consumers are strongly advised to meticulously examine their personal care items. Should any product align with the details of the recall, immediate disposal or return to the original point of purchase for a full refund is recommended. The Burkholderia cepacia complex can trigger symptoms such as fever and fatigue, and critically, it can lead to severe infections, particularly in individuals with compromised immune systems. Furthermore, direct application to skin lesions with contaminated products may result in localized infections.
For those who may have used any of the recalled topical treatments and subsequently experience signs of illness or infection, it is imperative to contact a healthcare professional without delay. Sedgwick is available to address public inquiries regarding this recall. They can be reached via telephone at 888-943-5190, operating Monday through Friday from 8 a.m. to 5 p.m. Eastern Time, or by email at dermarite5186@sedgwick.com.
This incident serves as a potent reminder of the critical need for robust regulatory oversight and stringent quality control in the manufacturing of consumer products, particularly those intended for personal care and health. From a public health perspective, such recalls, while disruptive, are absolutely essential for protecting the well-being of millions. It highlights how quickly and broadly potential dangers can spread through consumer goods, emphasizing the shared responsibility of regulatory bodies, manufacturers, and consumers themselves in maintaining safety standards. As a consumer, it reinforces the habit of conscientiously checking product information and staying informed about health alerts, reinforcing the idea that vigilance is a personal superpower in an increasingly complex global marketplace.
